
With valve-regulated lead-acid batteries(VRLA), you can store and transport energy easily. But some of that stored energy may ‘escape’ from the battery during the store. How can you stop such batteries ‘self-discharge? Let’s read on to find out.
Valve-regulated lead-acid batteries (VRLA) generate electricity owing to a chemical reaction inside the cell. In ideal, this happens when it is connected to a device that needs power. But the reaction could be happened at a smaller scale as well, while the battery’s electrodes are not connected. That’s meaning the battery’s charge gradually reduces over time. This phenomenon is called self-discharge. It’s perfectly normal.
VRLA battery self-discharge cannot be fully avoided. But it greatly depends on the battery type and its contents. Self-discharge refers to the electricity in the VRLA battery is consumed by itself, and self-discharge is inevitable. For a fully charged valve-regulated sealed lead-acid battery, if the capacity decreases by no more than 2% per day and night within 30 days, it is a normal discharge.
1. The electrolyte has a high specific gravity or the outside of the battery is not clean. If the electrolyte is spilled on the cover, it will cause leakage between the positive and negative poles, which will cause the valve-controlled sealed lead-acid battery to discharge.
2. The electrode separator of the valve-controlled sealed lead-acid battery is corroded, perforated and ruptured, causing a local short circuit or the active material falls off too much and deposit on the bottom of the battery so that the positive and negative plates are directly connected and short-circuited, causing the valve-controlled sealed lead-acid Self-discharge inside the battery.
3. The electrolyte is impure. The electrolyte contains harmful impurities (iron, manganese, arsenic, copper plasma), or distilled water is not added. At this time, the impurities in the electrolyte adhere to the electrode plate with the flow of the electrolyte, and a potential difference is formed, It will form many small batteries with self-contained paths inside the valve-regulated sealed lead-acid battery, making the valve-regulated sealed lead-acid battery in a short-circuit state. Tests have shown that if the electrolyte contains 1% iron, the valve-regulated sealed lead-acid battery will be fully discharged within 24-hours after being fully charged.
4. The valve-regulated sealed lead-acid battery plate is not pure, contains too much antimony, or contains harmful impurities, which will also form many small batteries. The potential difference between the impurity and the plate or between different impurities will become one. The local battery forms a loop through the electrolyte to generate a local current, which makes the valve-regulated sealed lead-acid battery self-discharge.
5. The valve-regulated sealed lead-acid battery has been stored for too long, and the water and sulfuric acid in the electrolyte are stratified owing to the different specific gravity, which makes the specific gravity of the electrolyte unevenly, forming a potential difference and self-discharge.
6. After the positive and negative plates are vulcanized, the pores of the separator are blocked, which causes the consumption in the battery to increase, and the valve-controlled sealed lead-acid battery produces self-discharge.
The self-discharge of the battery exceeds the normal value, which will cause unnecessary interference to the car’s travel, especially the reduction of the cruising range of pure electric vehicles, causing the breakdown phenomenon, which should be avoided.
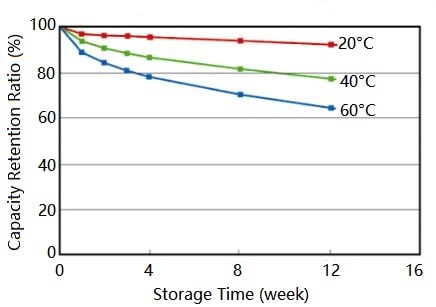
1.As with many chemical reactions, the rate and intensity at which it happens is influenced by the environment. Colder temperatures are generally good for the battery because we found the cold slows down the chemical reaction, thereby reducing any unwanted battery self-discharge. Therefore, the most logical thing is to seem to put the batteries in a freezer Contrarily you should avoid storing batteries in your freezer for a long time. The moist air in the freezers causes discharge too. Especially when you take your batteries out, condensation could damage them – causing them to no longer be able to use.
Store your batteries in a cool but dry place, preferable between 10 and 25°C. A few simple measures will keep unwanted battery self-discharge to a minimum. If you’re not sure about the batteries’ energy lev-el, you can always recharge them. That way, you’re sure your batteries are up for the task – and you can get the most out of your batteries day in day out.
2.Daily Maintenance
We should pay attention to the daily maintenance of the battery, check its cleanliness and integrity, and charge it in time when the power is less than half.
3. Be gentle in daily use
When installing or transporting the battery, please handle it gently to avoid contact between the wires in the battery and the outside, and also avoid connecting the external metal to the positive and negative poles.
4. Choose a battery with a good reputation
We should choose a battery on the market that have a good reputation, good quality, and great feedback from the market.

Valve-regulated lead-acid batteries (VRLA) self-discharge cannot be completely avoided. However, it greatly depends on the battery type and its quality.In short, VRLA batteries have self-discharge during storage and operation, which will lose part of the active material and increase the difficulty of maintaining battery capacity. The self-discharge rate is related to factors such as grid material, electrolyte density, active material purity and storage temperature.Spaceflight power’s VRLA batteries with low self-discharge, less than 2% per day, are your best choice!